Sorabh Kumar Agrawal*, Venkatachalam Karthikeyan
Department of Pharmacy, The Tamil Nadu Dr. MGR Medical University, Chennai, Tamil Nadu, India.
ORIGINAL RESEARCH ARTICLE
Volume 2, Issue 2, Page 95-100, May-August 2014.
Article history
Received: 7 August 2014
Revised: 10 August 2014
Accepted: 15 August 2014
Early view:20 August 2014
*Author for correspondence
E-mail: [email protected]
Aim: The aim of the present study was to evaluate a poly herbal syrup (PHS) including; powder microscopy, preliminary phytochemical screening and stability studies.
Material and methods: Powder microscopy and other pharmacognostic parameters like length and width of starch grains, length of trichome, ash value, and extractive value of individual drugs were performed to confirm the authenticity. Preliminary phytochemical screening was done by standard procedure. Formulation stability was determined by measuring its density, particle size and viscosity.
Results: The individual drug of the formulation was authenticated based on the powder microscopy, and other pharmacognostical parameter coincided with standard values. The preliminary phytochemical screening showed the presence of various chemical constituents in the individual drug. Stability studies showed PHS formulations were stable.
Conclusion: The results of the present study can be used for the genuine identification of the plant from its adulterants and subsequent screening for potent bioactivity in polyherbal formulations.
Keywords: Indigofera tinctoria, Inula racemosa, Jatropa curcas, Polyherbal Syrup, Pongamia pinnata, Rhaphidophora laciniata, Sapindus laurifolia, Terminalia chebula, Vitex negundo.
INTRODUCTION
Peptic ulcer is one of the major gastro‐intestinal disorders, which occurs due to an imbalance between the offensive (gastric acid secretion) and defensive (gastric mucosal integrity) factors. An estimated 15,000 deaths occur each year as a consequence of peptic ulcer (Palanivel et al., 2014). A number of drugs including H2 receptor antagonists and proton pump inhibitors are available for the treatment of peptic ulcer, but clinical evaluation of these drugs has shown incidence of relapse, side effects and drug interactions (Srinivas et al., 2013). This has been the rationale for the development of new antiulcer drugs and search for novel molecules has been extended to herbal drugs that offer better protection and decreases relapses. Phytogenic agents have traditionally been used by herbalists and indigenous healers for the preventive and treatment of ulcer. A botanical compound with antiulcer activity includes flavonoids, saponins, tannins, gums and mucilage (Venkateswararao and Venkataramana, 2013).
Flavonoids have been reported to act in the gastrointestinal tract, having anti-secretory (Di Carlo et al., 1999), antiulcer, antioxidant properties (La Casa et al., 2000) and cytoprotective (Borrelli and Izzo, 2000; Btista et al., 2013). They protect the gastric mucosa against a variety of ulcerogenic agents via several mechanisms of action, mainly free-radical scavenging and antioxidant properties, increased mucus production, antisecretory action, and inhibition of the Helicobacter pylori growth (de Lira Mota et al., 2009).
Polyphenols display a number of pharmacological properties in the GIT area, acting as antisecretory, cytoprotective, and antioxidant agents (Sumbul et al., 2011). Polyphenolic compounds have also been reported to have a beneficial role in gastric ulcers, as it has been suggested that phenols stimulate PGE2 formation (Ismail et al., 2012; Lakshmi et al. 2014). Gastro‐protective effect of saponin is due to activation of mucous membrane protective factors and triterpenoid saponin regards to the plant for antiulcer activity (Yoshikawa et al., 2005). Tannins prevent ulcer development due to their protein precipitating and vasoconstricting effects (Awaad et al., 2013). Their astringent action can help to precipitate microproteins on the ulcer site, thereby, forming an impervious layer over the lining, which hinders induced gastric ulcer in rats, as evidenced by the gut secretions, and protects the underlying mucosa from reduction in the ulcer scores (Nwafor et al., 2000). Many drugs obtained from natural source, which are used in allopathy and all other indigenous system of medicine for various ailments. Now a day, the polyherbal formulation is becoming popular for various longstanding diseases just like combination therapy. Many studies have been carried out and many formulations are also commercially available with remarkable protection and acceptance.
In the present study, we planned to formulate polyherbal formulations with various traditionally using crude drugs based on the ethnopharmacological use, such as Indigofera tinctoria, Terminalia chebula, Jatropa curcas, Raphidophora laciniata, Inula racemosa, Pongamia pinnata, Vitex nedungo and Sapindus laurifolia (Motamarri N et al., 2013; Sharma et al., 2012, Sivarajan and Indira, 1994; Amin et al., 2013; Chopade et al., 2008; Telang et al., 1999; Ali et al., 1998; Singh and Kshirsagar, 2001, respectively) into two different concentrations and different base. Further, stability studies of both the formulations were also performed.
MATERIALS AND METHODS
Chemicals: Formalin, acetic acid, ethyl alcohol, chloral hydrate, toludine blue, phloroglucinol, glycerin, hydrochloric acid and all other chemicals used in this study were of analytical grade.
Plant collection and authentication: All plant specimens was identified and authenticated by TNAU, Botanical Survey of India, Coimbatore, Tamil Nadu, India.
Macroscopic analysis: Macroscopic study of the plant was performed. The shape, size, surface characters, texture, color, odor, taste etc. were noted. It is a technique of qualitative evaluation based on the study of morphological and sensory profiles of herbs (Agrawal et al., 2014).
Physicochemical analysis: Total ash, acid insoluble ash, water soluble ash, loss on drying, extractive values were performed as per standard procedure (Agrawal and Paridhavi, 2007; Ahmad et al., 2007).
Micrscopical parameters: Microscopical parameters such as length and width of fibres, length of trichomes, and diameter of starch grains were determined.
Preliminary phytochemical screening: Preliminary phytochemical screening was carried out to find out the presence of various phytoconstituents using standard procedure (Khandewal, 2008; Rangari, 2003).
Powder microscopy: Coarse powder was used to study the microscopical characters (Purvis et al., 1966).
Formulation of antiulcer syrup: After microscopic evaluation, all eight drugs were converted into coarse powder form. Coarse powders were separately sieved through 100 mesh. The two different formulations were prepared using same drug in different base mentioned (Table 1 to 3).
RESULTS
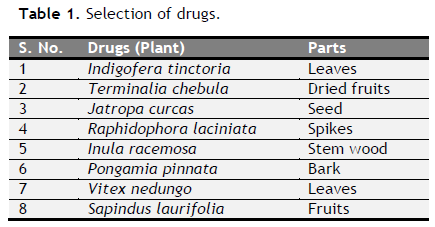 |
Table 1. Selection of drugs. Click here to view full image |
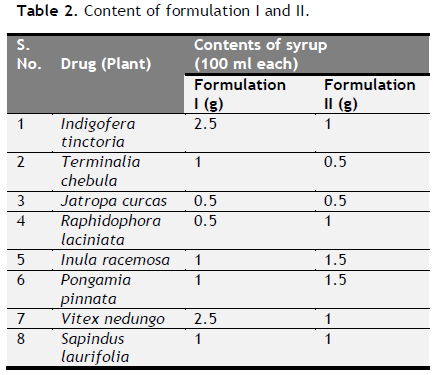 |
Table 2. Content of formulation I and II. Click here to view full image |
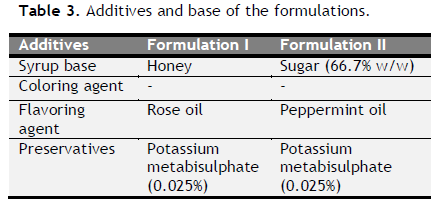 |
Table 3. Additives and base of the formulations. Click here to view full image |
Evaluation of antiulcer formulations (syrup)
Determination of density: The density of the formulation was determined by using pyknometer. The capacity of pyknometer was noted. The volume and weight of the antiulcer formulation F1 and F2 were compared with same volume and weight of water.
The density of liquid = weight of liquid/ weight of water X density of water
Determination of particle size: The particle size drugs presence in the formulation were determined using eye piece and stage micrometer. Eye piece micrometer was calibrated by using stage micrometer. A small quantity of each formulation was spread in the slide and the number of division covered by the each individual particle in the eye piece micrometer was noted. From the above value the minimum, average and maximum diameter of particle present in the formulation were calculated.
Determination of Sedimentation rate: Formulation F1 and F2 were taken in 50 ml graduated measuring cylinder separately up to 50 ml mark. The measuring cylinder was shaken for 15 minutes and set aside. After every 15 minutes sedimentation volume was measured and tabulated up to 1 hour.
Determination of Viscosity: The viscosity of the formulation was determined by using Oswald’s viscometer. Initially the density of the formulation F1 and F2 were determined. Then the time taken to for the flow of liquid and water through Oswald’s viscometer from the upper level to the bottom of the bulb were noted. The viscosity is calculated by using the following formula.
Viscosity of the formulation = Þ1t1/Þ2t2 ×ʼnwŊ
Macroscopy
All eight drugs in this poly herbal formulation were authenticated by means of macroscopical analysis. Eight drugs (colour, odour, taste and shape) were coincided with standard drug:
Indigofera tinctoria
Part: dried leaves; color-green, grayish black; shape-ovate or lanceolate; apex-acute; base-obtuse; margin-entire; venation-palmate; texture-coriaceous; lamina or surface-glabrous. Powder form: color-pale green; odor-leafy odor; taste-bitter.
Terminalia chebula
Part: dried fruits; color-yellow to orange brown; size-3.75 cm; shape-ellipsodial or ovoid; types-trupes; surface-glabrous, shining; seeds-hard, yellow; powder form-color-light brown; odour-mucilaginous; taste-sweet, astringent.
Jatropa curcas
Part: seeds; color-brownish black, dull brown; type-split into three parts; surface-glabrous, shining. Powder form: color-pale grey; odor-fatty odor; taste-acrid and sweet.
Rhaphidophora laciniata
Part: dried fruits; color-bluish brown; shape-large, ovoid-oblong, erect, solid fleshy spikes; type-spikes, size-longer, 4 to 8.5 cm. Powder form: color-dark brown black; odor-aromatic; taste-pungent and bitter.
Inula racemosa
Part: stem wood; type-sapwood; shape-cylindrical; size-6-8 cm in length, 2-3 cm in diameter; outer surface-covered with gray color bark; inner surface-straw yellow color and fissured; Powder form: color-yellow to light brown; odor-odourless; taste-astringent.
Pongamia pinnata
Part: dried bark; size & shape-2 to 3 in length, 1.5 – 2.5 cm in width, curved, single squill with annulated markings; outer surface-greyish brown with yellow markings; inner surfaces-light brown to yellowish brown; powder form; color-brown; odor-slightly aromatic; taste-sweet and mucilaginous.
Vitex negundo
Part: dried leaves; color-brownish black, dull brown; size & shape-lanceolate, estipulate, long petiole, trifoliate leaflets 3-5 cm long & 1-1.5 cm wide; apex-acuminate; base-acute; margin-entire; texture-membranous; surface-glaborous; powder form; color-bluish green, greyish black; odor-leafy; surface-glaborous.
Sapindus laurifolia
Part: dried fruits; color-yellowish brown to reddish brown; seed-black; bulp-saponaceous; size & shape-round, 2-3 cm in diameter; outer surface-wrinkled. Powder form; color-brown; odor-odorless; taste-mucilaginous & bitter.
Physicochemical analysis
Total ash, acid insoluble ash, water soluble ash, loss on drying, extractive values and crude fibre content were determined and tabulated (Table 4 to 6). Foreign organic matter of all varieties was nil. Physiochemical analysis (Total ash, acid insoluble ash, water soluble ash, loss on drying, extractive values and crude fibre content) of all eight drugs were showed its genunity. All values were coincided with standard one.
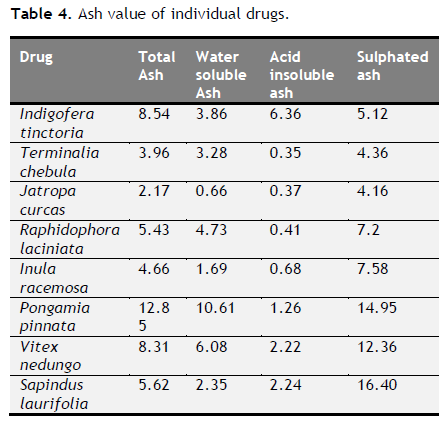 |
Table 4. Ash value of individual drugs. Click here to view full image |
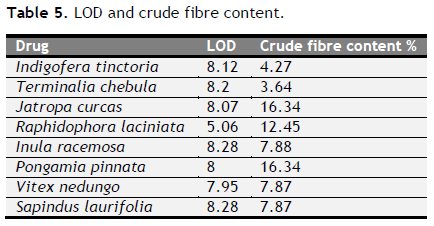 |
Table 5. LOD and crude fibre content. Click here to view full image |
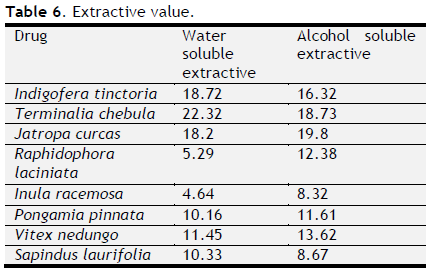 |
Table 6. Extractive value. Click here to view full image |
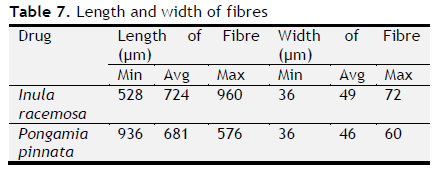 |
Table 7. Length and width of fibres. Click here to view full image |
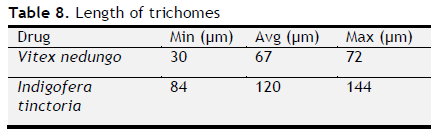 |
Table 8. Length of trichomes. Click here to view full image |
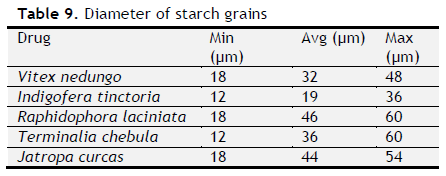 |
Table 9. Diameter of starch grains. Click here to view full image |
Preliminary phytochemical screening
All crude drug powder was subjected to chemical test for identification of its active constituents and results are tabulated (Table 10). Preliminary phytochemical screening showed presence of flavonoids, phenolic compound and saponins in most of the individual drug. Antiulcer effect may be due to the presence of flavonoids, phenolic compound and saponins.
Powder microscopy
All crude drug powder was subjected to powder microscopic evaluation after staining with phloroglucinol and conc.HCL and individual power characters were shown.
Indigofera tinctoria: trichomes, stomata with epidermis and calcium oxalate crystal sheath.
Terminalia chebula: stone cells, pieces of lignified trichome, epidermis with parenchyma and endosperm.
Jatropa curcas: pigment layer, oil globules, sclrenchymatous layer and epidermis with oil glands.
Raphidophora laciniata: parenchyma, endocarp, unlignified fibres and starch grains.
Inula racemosa: cork, fibres, starch grains and xylem with parenchymatous cell.
Pongamia pinnata: phloem parenchyma, cork, stone cells and phloem parenchyma with calcium oxalate crystal.
Vitex nedungo: trichomes, stomata, calcium oxalate crystal and vessels.
Sapindus laurifolia: endosperm with calcium oxalate crystal, epicarp, trichomes, and perisperm with calcium oxalate crystal.
Powder microscopical character of all drugs of PHS was coincided with standard.
Evaluation of antiulcer syrup:
Density, particle size, sedimentation rate and viscosity were determined and tabulated (Table 11 to 14). Formulation I and II had density, particle size, sedimentation rate and viscosity within the limit of the standard syrup.
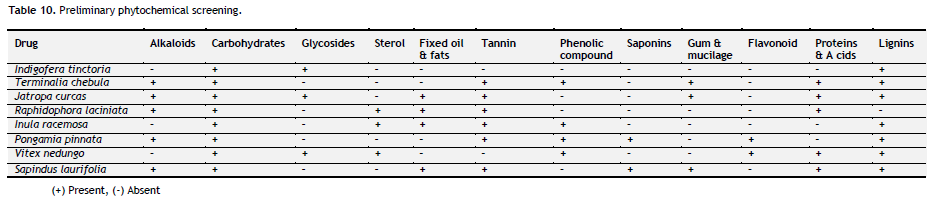 |
Table 10. Preliminary phytochemical screening. Click here to view full image |
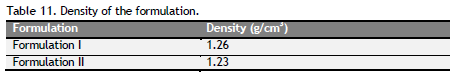 |
Table 11. Density of the formulation. Click here to view full image |
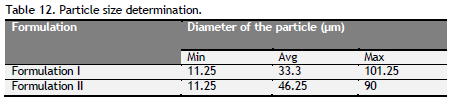 |
Table 12. Particle size determination. Click here to view full image |
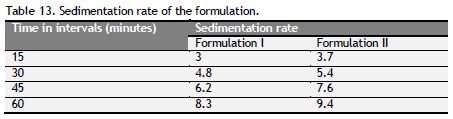 |
Table 13. Ash value of individual drugs. Click here to view full image |
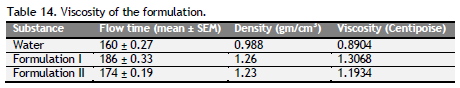 |
Table 14. Sedimentation rate of the formulation. Click here to view full image |
DISCUSSION
Sensory evaluation plays a vital role in determining the suitability of a crude drug. Organoleptic evaluation of a crude drug is mainly for qualitative evaluation based on the observation of sensory and morphological profile (Periyanayagam and Karthikeyan, 2013). In this report, various morphological, microscopical, physicochemical standards have been developed. Hence we have undertaken this study to serve as a tool for developing standards for identification, purity and quality of various crude drugs for antiulcer formulation.
Adulteration and misidentification of crude drugs can cause serious health problems to consumers and legal problems for the pharmaceutical industries. It can be conducted via a variety of techniques, namely macro and microscopic identification and chemical analysis especially description of microscopic botanical aspects to determine definitively the proper species of plant material while it is still in its non extracted form (Karthikeyan et al., 2014). The observation of cellular level morphology or anatomy is a major aid for the authentication of drugs. These characters are especially important for identification of powdered drugs, because in these cases most of the morphological diagnostic features are lost (Aruna et al., 2014). Microscopic evaluation is one of the simplest and cheapest methods for the correct identification of the source of the materials (Periyanayagam et al., 2013). The macroscopic and organoleptic characters of the leaf can serve as diagnostic parameters (Agrawal et al., 2014). Macroscopic and organoleptic characters of all eight drugs conform its identity and purity.
The ash values are particularly important to find out the presence or absence of foreign inorganic matter such as metallic salts and or silica (Aruna et al., 2014). Acid insoluble ash provides information about non-physiological ash produced due do adherence of inorganic dirt, dust to the crude drug. Increased acid insoluble ash indicates adulteration due do dirt, sand (or) soil. The extractive values are primarily useful for the determination of exhausted or adulterated drug and helpful in the detection of adulteration (Periyanayagam et al., 2013). Ash value, extractive value and LOD were done to confirm the authenticity. Phytochemical evaluation and molecular characterization of plants is an important task in medicinal botany and drug discovery. Preliminary phytochemical screening was done by standard procedures to found out chemical constituents. Preliminary phytochemical screening showed the presence of various chemical constituents in the individual drug Powder microscopy of individual drugs and other pharmacognostic parameters like length and width of starch grains, length of trichome and diameter of starch grains were determined. Based on the powder microscopy the individual drug of the formulation was authenticated and other pharmacognostical parameter coincided with standard value. A stability study was carried out to check the stability of the formulations. Stability of this formulation was determined by measuring its density, particle size and viscosity. Stability studies showed PHS formulation both I and II were stable.
CONCLUSION
The results of the present study can be used for the genuine identification of the plant from its adulterants and subsequent screening for potent bioactivity in polyherbal formulations.
CONFLICT OF INTEREST
None declared.
REFERENCES
Agrawal SK, Karthikeyan V, Parthiban P. Pharmacognostic and Physiochemical standardization of the leaves of Scoparia dulcis L. Int J Univers Pharm Bio Sci. 3, 131-144, 2014.
Agrawal SS, Paridhavi M. Herbal Drug Technology, 2nd edition. Universities press Pvt. Ltd: Hyderabad, India, pp. 626-644, 2007.
Ahmad I, Aqil F, Owais M. Modern Phytomedicine-Turning medicinal plants into drugs. John Wiley and Sons: Weinheim, pp. 25-57, 2007.
Ali MS, Ahmad VU, Azahar I, Usmanghani K. Some medicinally important plant and their uses. Hamdard Medicus 41, 96-102, 1998.
Amin S, Kaloo ZA, Singh S, Altaf T. Medicinal importance of genus Inula- A review. Int J Cur Res Rev. 5, 20-26, 2013.
Aruna A, Vijayalakshmi K, Karthikeyan V, Nandhini SR, Jegadeesh S. Water Melon Plant (Citrullus lanatus): Pharmacognostical Standardization and Phytochemical Screening of its Leaves. Pharmanest 5, 2184-2191, 2014.
Awaad AS, El-Meligy RM, Soliman GA. Natural products in treatment of ulcerative colitis and peptic ulcer. J Saudi Chem Soc. 17, 101–124, 2013.
Borrelli F, Izzo AA. The plant kingdom as a source of anti-ulcer remedies. Phytother Res. 14, 581-591, 2000.
Chopade VV, Tanker AN, Pande VV, Tekade AR, Gowekar NM, Bhandari SR et al., Pongamia pinnata: Phytochemical constituents, traditional uses and pharmacological properties: A review. Int J Green Pharm. 2, 72-75, 2008.
de Lira Mota KS, Dias GEN, Pinto MEF, Luiz-Ferreira Â, Souza-Brito ARM, Hiruma-Lima CA, Barbosa-Filho JM, Batista LM. Flavonoids with gastroprotective activity. Molecules. 14, 979-1012, 2009.
Di-Carlo G, Mascolo N, Izzo AA, Capasso F. Flavonoids: Old and new aspects of a class of natural therapeutic drugs. Life Sci. 64, 337-353, 1999.
Ismail M, Rahman S, Zada A, Abbas M, Ali T, Niaz U. Analgesic, anti GIT motility and toxicological activities of Pistacia integerrima Stewart ex Brandis bark in mice. J Medicinal Plants Res, 6, 2827-2831, 2012.
Karthikeyan V, Agrawal SK, Parthiban P, Nandhini SR. Multivitamin plant: pharmacognostical standardization and phytochemical profile of its leaves. J Pharm Res. 8, 910-915, 2014.
Khandewal KR. Practical Pharmacognostic Techniques and Experiments. 19th edition. Nirali Prakashan: Pune, India, pp.149-156, 2008.
La Casa C, Villegas I, Alarcon DL, Motilva V, Martin MJ. Evidence for protective and antioxidant properties of rutin, a natural flavone, against ethanol induced gastric lesions. J Ethnopharmacol. 71, 45–53, 2000.
Lakshmi BVS, Muvvala S, Shashank P. Protective effect of Vitis vinifera in doxorubicin-induced oxidative stress in rats- A preliminary study. Int J Adv Pharmacy Med Bioallied Sci. 2, 7-14, 2014.
Motamarri NS, Karthikeyan M, Rajasekar S, Gopal V. Indigofera tinctoria Linn - A Phytopharmacological Review. Int J Res Pharm Biomed Sci. 3, 164-169, 2012.
Nwafor PA, Okwuasaba FK, Binda LG. Anti diarrheal and anti ulcerogenic effects of methanolic extract of Asparagus pubescens root in rats. J Ethnopharmacol. 72, 421–427, 2000.
Palanivel, Agrawal SK, Manjulvani, Senthil Kumar K, Karthikeyan V, Periyasamy P, Sivakumar GM. Evaluation of anti ulcer activity of Pithecellobium dulce (seeds) in rats using pylorus ligation. World J Pharmacy Pharmaceut Sci. 3, 2175-2184, 2014.
Periyanayagam K, Karthikeyan V. Pharmacognostical, SEM and XRF profile of the leaves of Artocarpus heterophyllus Lam. (Moraceae) – a contribution to combat the NTD. Innovare J Life Sci. 1, 23-28, 2013.
Periyanayagam K, Sasikala T, Karthikeyan V, Jegadeesh S, Jancy Gracelet R. Pharmacognostical and phytochemical studies on the leaves of Anacardium occidentale Linn. World J Pharm Sci. 2, 41-48, 2013.
Purvis MJ, Collier DC, Wallis D. Laboratory Techniques in Botany, 2nd edition. Butter worth & Co Ltd: UK, pp. 82 -169, 1966.
Rangari VD. Pharmacognosy and Phytochemistry. 1st editon. Career Publication: Nashik, India, pp. 224, 2003.
Sharma S, Dhamija HK, Parashar B. Jatropha curcas: A Review. Asian J Res Pharm Sci. 2, 107-111, 2012.
Singh NP, Kshirsagar RD. Some less known ethano medicinal uses from Mysore and Coorg districts, Karnataka. Ancient Sci Life. 20, 20-25, 2001.
Sivarajan VV, Indira B. Ayurvedic drugs and their plant sources. Oxford and IBH publishing Pvt Ltd: New Delhi, India, 1994.
Srinivas TL, Lakshmi SM, Shama SN, Reddy GX, Prasanna KR. Medicinal plants as anti-ulcer agents. J Pharmacog Phytochem. 2, 91-97, 2013.
Sumbul S, Ahmad MA, Mohd A, Mohd A. Role of phenolic compounds in peptic ulcer: An overview. J Pharm Bio allied Sci. 3, 361-367, 2011.
Telang RS, Chattergie S, Varshnega S. Studies on analgesic and anti-inflammatory activity of Videx nedungo Linn. Indian J Pharmacology 31, 363-366, 1999.
Venkateswararao C, Venkataramana K. A pharmacological review on natural antiulcer agents. J Global Trends Pharmaceut Sci. 4, 1118-1131, 2013.
Yoshikawa M, Morikawa T, Ning L, Nagatomo A, Xian L, Matsuda H. Bioactive Saponins and Glycosides. XXIII. Triterpene Saponins with Gastroprotective Effect from the Seeds of Camellia sinensis- Theasaponins E3, E4, E5, E6, and E7. Chem Pharm Bull. 53, 1559-1564, 2005.








 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.